Artemisian r&d
Our objectives are:
to create products from the medicinal herb Artemisia annua and Artemisia afra with markets in developing countries and elsewhere to capitalize on:
– the standardization of plant cultivation to meet GAP certification
– quality of assurance in manufacturing via GMP certification
– the global need to address ‘natural product’ efficacy against tropical maladies. *
Our ongoing program:
To propagate, harvest and manufacture GRAS products of Artemisia annua L. and Artemisia afra, centralized at our existing horticultural nursery and processing facility in South Florida. All of the medicinal herbs grown, harvested and milled by Atelier Temenos at our Homestead, FL site have been approved and listed on the NOP Organic Input Review Registry
The uniqueness of our existing program is:
The quality control and standardization for propagation, harvesting and manufacturing via authorized certifications of a 100% Artemisia based product. There are no Artemisia annua or Artemisia afra commercial products that meet that criteria globally.
*An estimated 219 million people suffered from malaria in 2017 and 435,000 died. More than 90 % of the deaths were in Africa and over 60% were among children under 5-year-old. These deaths are predominantly in developing countries, located in tropical climates…. however, with the rise in global warming, there are predictions that even more humans will become venerable to malaria outbreaks.
The active ingredient artemisinin synthesized from the A.annua and used as artemisinin combination therapy (ACT), is the current best therapeutic for treating malaria. But it is expensive to produce, is in short supply in areas hit hardest by the disease, and the malaria parasite is showing signs of resistance to ACT, particularly in Southeast Asia.
Today, it has been proven that dried leaves of the A. annua plant delivers 40 times more artemisinin in the blood than does the drug based on the chemical extract of the plant, that it is more effective in knocking out the parasite and reduces the level of parasite infection. It constitutes a robust natural combination therapy, one whose benefits far surpass those of ACT and other combination drugs.
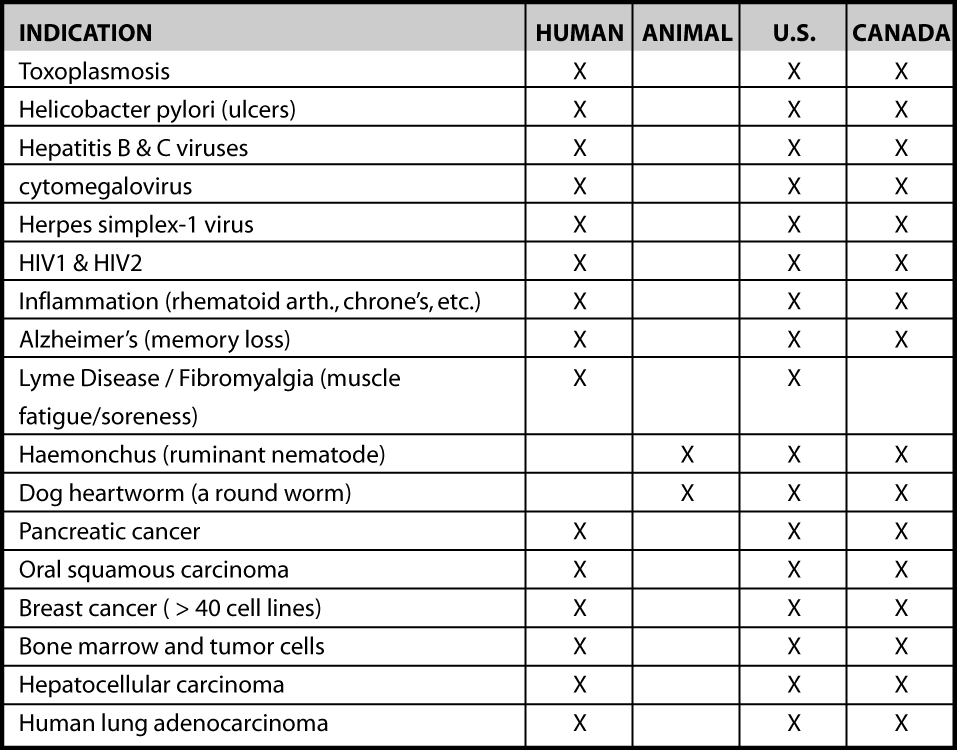
Artemisia annua is also known to be efficacious against a range of other disease, including other tropical maladies. Recent WPI research includes positive tuberculosis efficacy. The below table lists areas where artemisinin many be beneficial based upon current data available.
To meet federal regulations in order to enter the pharmaceutical drug market. The list of targeted indications is shown below in Figure 1. Market analysis for both short and long term will include both the United States and Canada.